 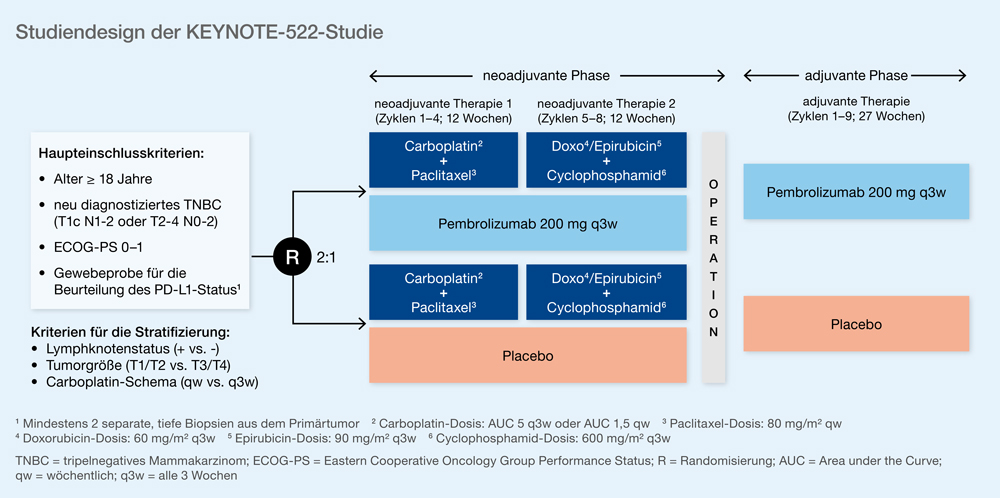
Of these patients, 104 (76.5%) in the pembrolizumab plus chemotherapy group and 60 (75.0%) in the placebo plus chemotherapy group had a tumor PD-L1 combined positive score of 1 or greater. Results A total of 216 of 1174 randomized patients (all female median age, 46.0 years) were from Korea, Japan, Taiwan, and Singapore (136 in the pembrolizumab plus chemotherapy group and 80 in the placebo plus chemotherapy group). Main Outcomes and Measures The main outcome was pCR (no evidence of primary tumor after neoadjuvant therapy or carcinoma in situ after neoadjuvant therapy and no regional lymph node involvement after neoadjuvant therapy) at the time of definitive surgery and EFS. After definitive surgery, patients received pembrolizumab or placebo every 3 weeks for 9 cycles or until recurrence or unacceptable toxic effects. Intervention Patients were randomized 2:1 to 4 cycles of pembrolizumab (200 mg every 3 weeks) or placebo plus carboplatin and paclitaxel and another 4 cycles of pembrolizumab or placebo plus doxorubicin or epirubicin and cyclophosphamide before surgery. This secondary analysis included adults enrolled in Asia with newly diagnosed, previously untreated, nonmetastatic triple-negative breast cancer (tumor stage T1c and nodal stage N1-2 or tumor stage T2-4 and nodal stage N0-2) and Eastern Cooperative Oncology Group performance status of 0 to 1, regardless of programmed cell death ligand 1 (PD-L1) status. Data cutoff for pCR analysis was September 24, 2018. For interim EFS and overall survival (OS) analyses (data cutoff, March 23, 2021), median follow-up was 39.8 months (range, 30.4-46.9 months) for pembrolizumab plus chemotherapy and 40.8 months (range, 30.1-46.9 months) for placebo plus chemotherapy. Objective To evaluate efficacy and safety outcomes for patients enrolled in East/Southeast Asia (Asia) in KEYNOTE-522.ĭesign, Setting, and Participants KEYNOTE-522, a multicenter, double-blind, randomized clinical trial, enrolled 1174 patients between March 7, 2017, and September 13, 2018. 
Importance In the phase 3 KEYNOTE-522 study, addition of pembrolizumab to neoadjuvant chemotherapy followed by adjuvant pembrolizumab significantly increased pathologic complete response (pCR) and event-free survival (EFS) vs neoadjuvant chemotherapy in patients with early triple-negative breast cancer. Shared Decision Making and Communication.Scientific Discovery and the Future of Medicine.Health Care Economics, Insurance, Payment.Clinical Implications of Basic Neuroscience.Challenges in Clinical Electrocardiography.  
These works are owned by Magdalen Medical Publishing (MMP) and are protected by copyright laws and treaties around the world. This interview took place at the European Society for Medical Oncology (ESMO) 2023 Congress in Madrid, Spain. The benefit with pembrolizumab was consistent across pre-specified subgroups. The 5-year follow up data confirm these earlier findings: the 60-month EFS rates were 81.3% in the pembrolizumab and chemotherapy arm, and 72.3% in the chemotherapy and placebo arm. It was previously reported that this pembrolizumab regimen resulted in statistically significant improvements in pathological complete response (pCR) and event-free survival (EFS). Peter Schmid, FRCP, MD, PhD, Barts Cancer Institute, London, UK, discusses 5-year follow-up data from the Phase III KEYNOTE-522 (NCT03036488) trial which aimed to investigate pembrolizumab plus chemotherapy as neoadjuvant therapy, and pembrolizumab as adjuvant therapy, in patients with triple-negative breast cancer. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
